JAMA CONNECT--Case study--Medical Devices
Accelerate the Development of Safe and Effective Medical Devices & Life Science Products with Jama Connect
In the medical device & life sciences industries, the transformation from hardware-focused devices to diverse products driven by software — in combination with hardware and bioware — is reshaping the tools and processes by which devices and related products are developed. Jama Connect for medical device requirements management is purpose-built to reduce the effort required to achieve regulatory compliance by managing design controls for device requirements and related risks, simplifying regulatory submissions and audit preparations, and accelerating time to market.
With Jama Connect You Can:
· Efficiently manage requirements, risks, verification, and validation through Live Traceability™
· Quickly align systems engineering disciplines across hardware, software, and bioware product development teams
· Simplify regulatory submissions and compliance with industry standards
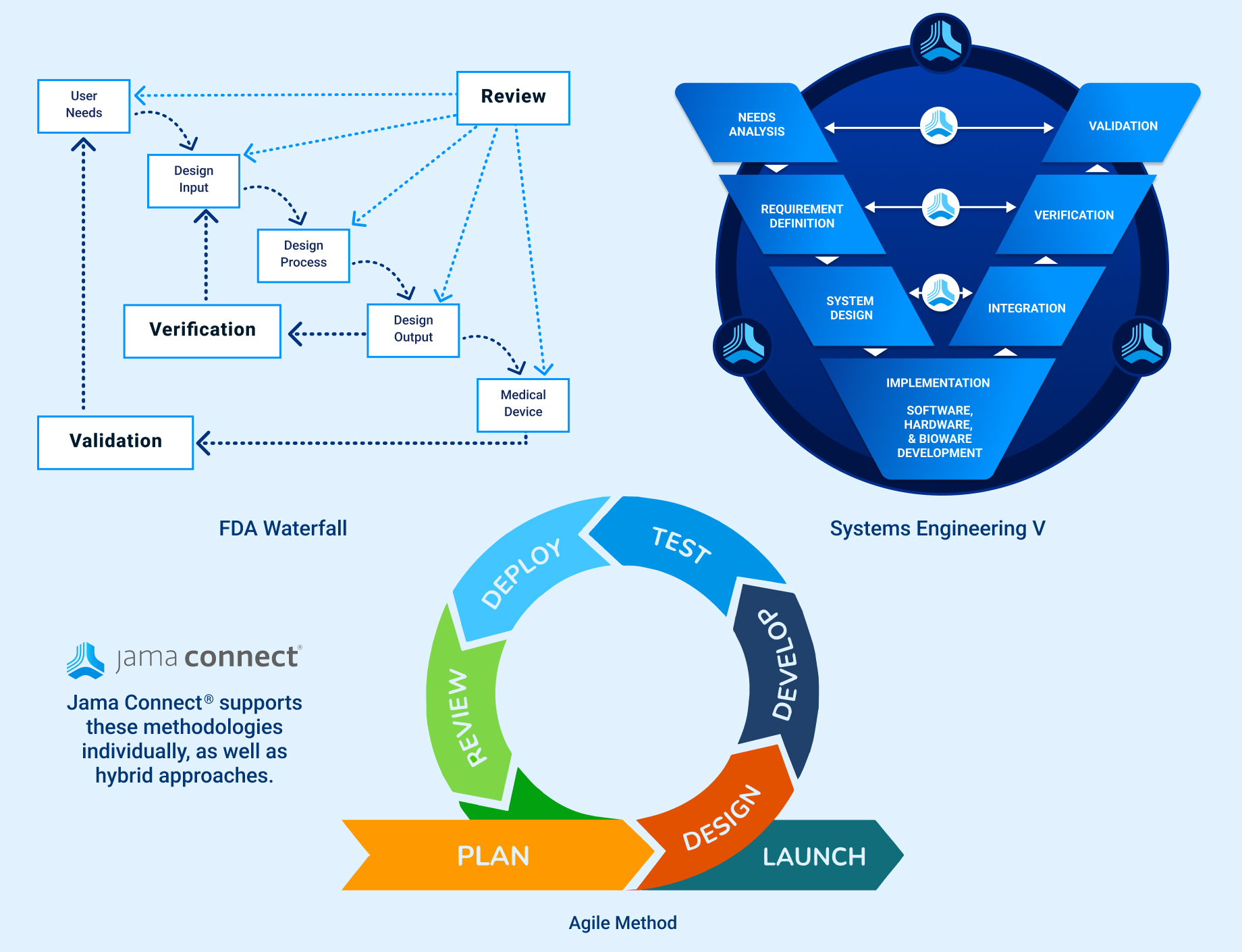
Jama Connect provides a flexible solution to manage all of your product development methods.
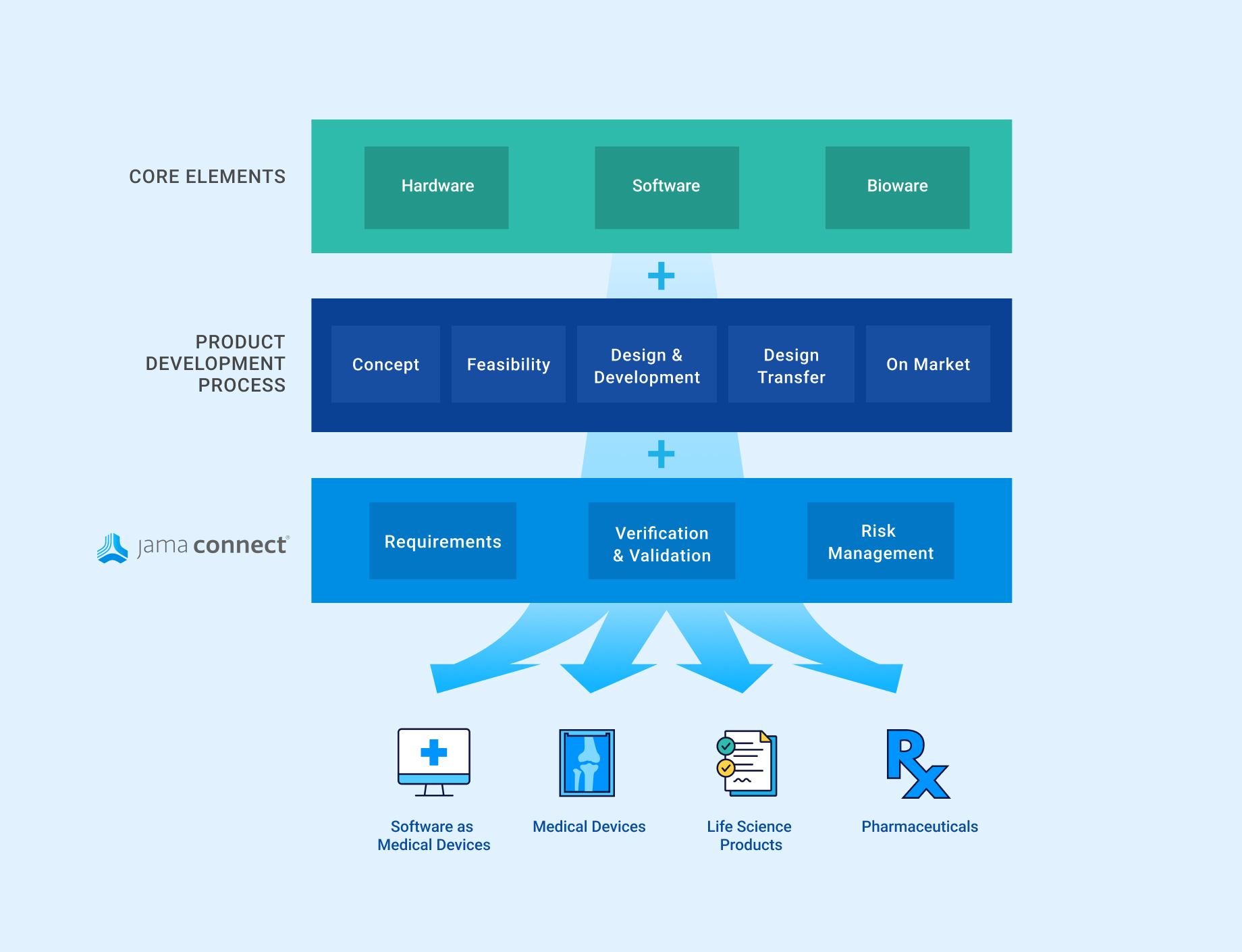
Jama Connect offers a comprehensive solution for simplifying the product development process across the device, product, system and software lifecycle.
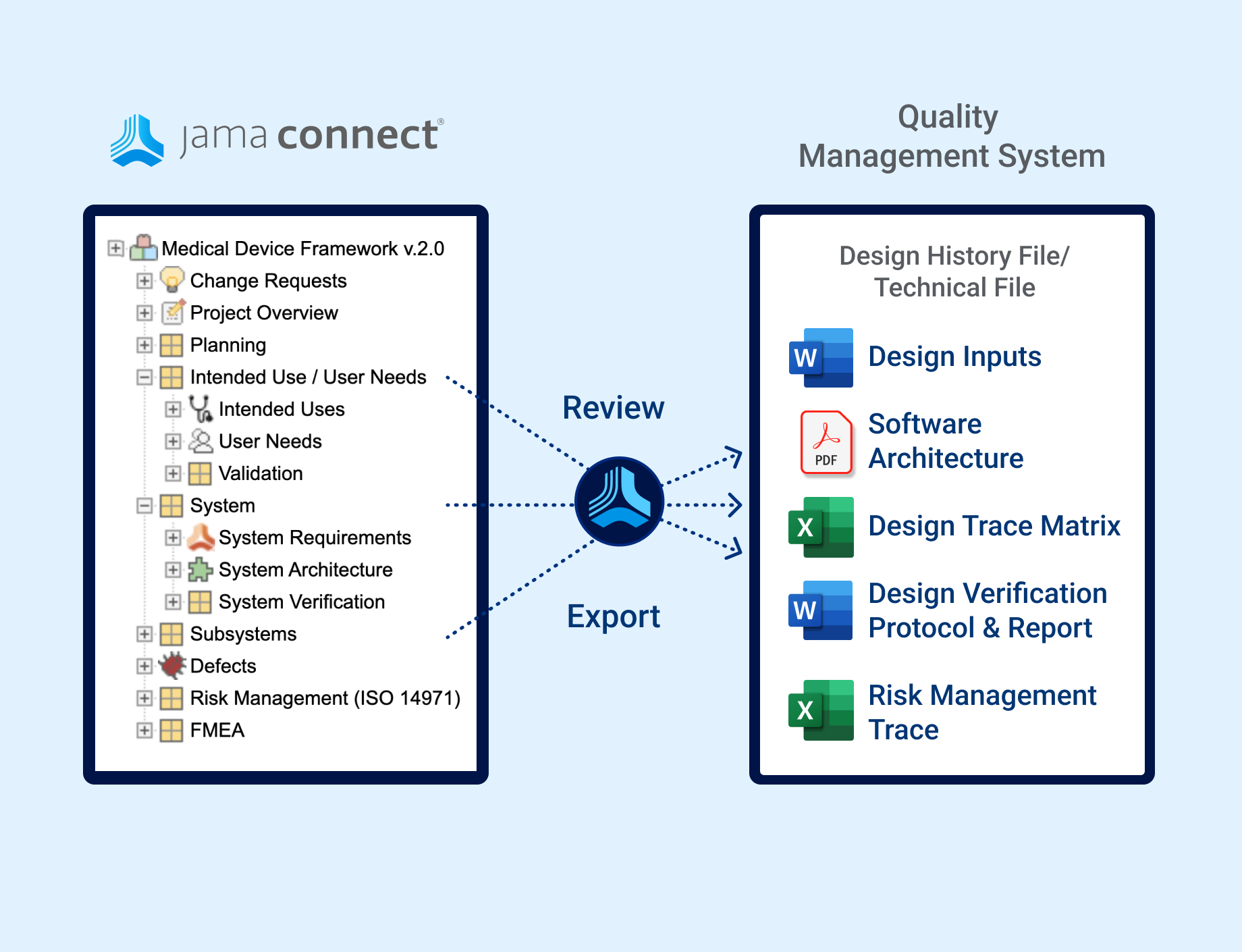
Jama Connect streamlines review cycles and document creation for your design history file/technical file to complement your quality management system.
“Jama Connect’s automation allows us to efficiently manage each release. The tool allows us to reduce manual processes involved in generating traceability and focus our efforts on reviews of the trace matrix. Efficiency and speed are critical in managing our software cadence and to be competitive in the marketplace.”
Sandhya MitnalaHead of Quality and Regulator
“With Review Center, we review things more incrementally, and we can catch changes, or things to fix, very early in the process. So, the cost at the end is less.”
Carmen PazosDiagnostic Divisions R&D, Instruments Sr Manager
Jama Connect for Medical Device & Life Sciences Development is a single powerful platform for medical device teams to manage design controls for device requirements and related risks, simplifying regulatory submissions and audit preparations while accelerating time to market. It is built to include standard medical device frameworks that support key medical device software standards and regulations, including:
· Regulatory Agencies: FDA 21 CFR 820.30, 21 CFR 11, EU MDR, EU IVDR
· Quality Systems: ISO 13485
· Risk Management Process and Methods: ISO 14971, IEC 60812
· Medical Software Development: IEC 62304, AAMI TIR 45 and SW 96
· Human Factors and Usability Engineering: HE 75, IEC 62366
Areas within the medical device/life sciences space Jama Software supports include Medical Devices, Diagnostics (IVDs, LDTs, RUOs), Medical Device Software, Pharmaceuticals, Life Sciences, and more!
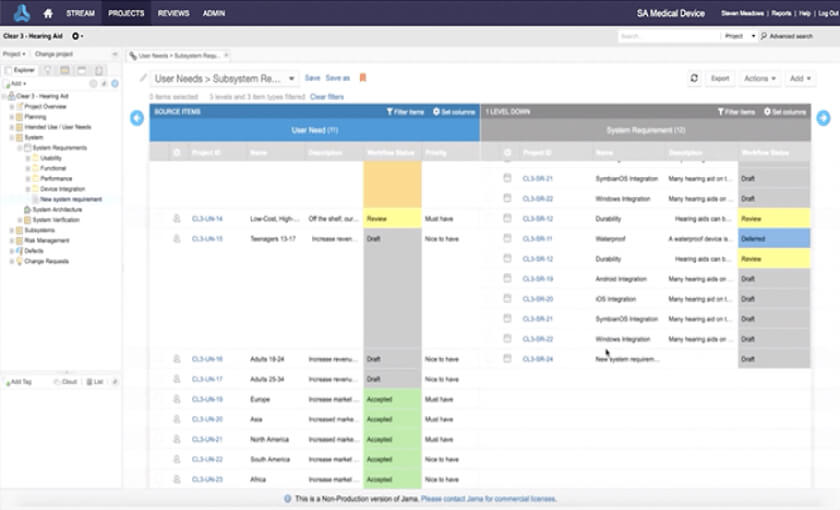
Link high-level requirements to more detailed and sub-system requirements to assure proper validation before release.
• Increase efficiency
• Drive alignment
• Mitigate organizational risk
Write and maintain requirements showing links to related upstream and downstream requirements
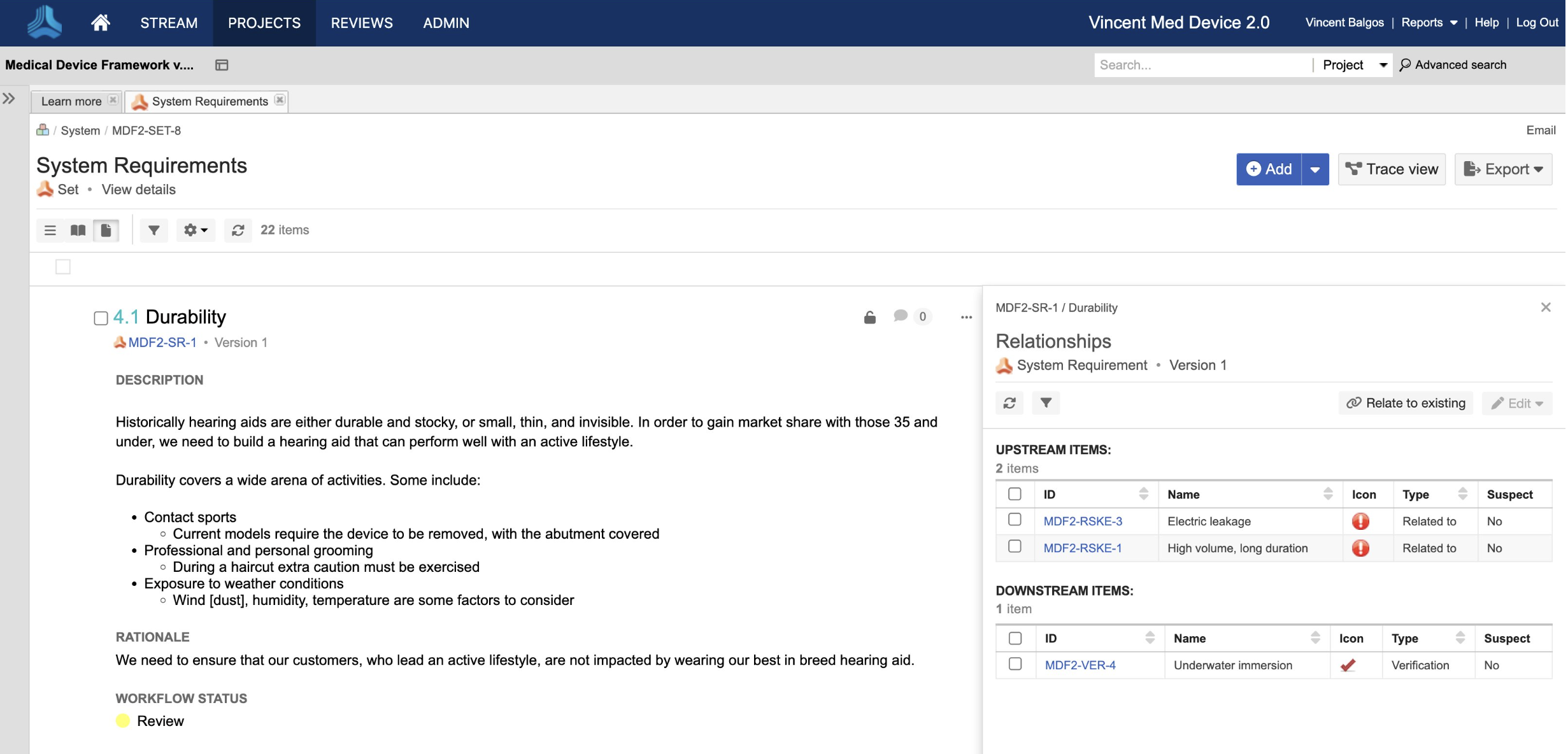
Establish a medical device risk management framework to ensure quality and minimize design changes.
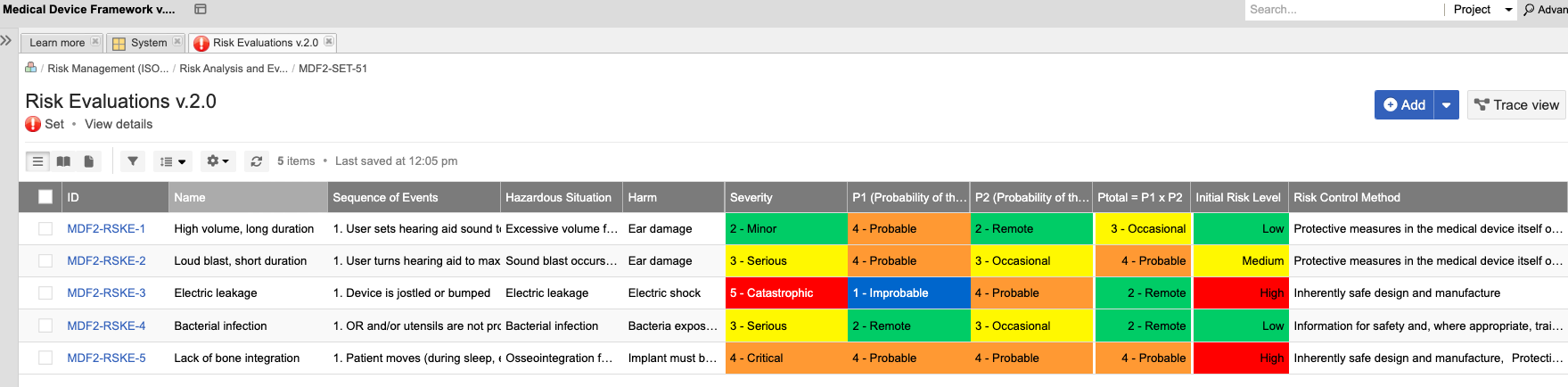
• Align with ISO 14971:2019
• Mitigate risk earlier in development
• Support the path to regulatory compliance
Seamlessly manage traceability to test cases used for verification and validation.
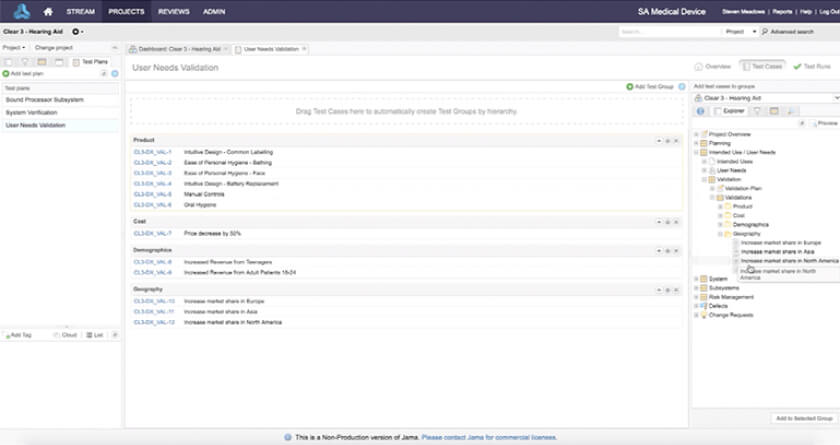
• Provide evidence to comply with regulations and standards, like 21 CFR Part 820.30
• Manage traceability to test cases used for verification and validation
Reuse common items with their relationships from the library or another project and sync to view differences over time.

Achieve compliance with FDA 21 CFR Part 11 when you combine with your organization’s quality process.
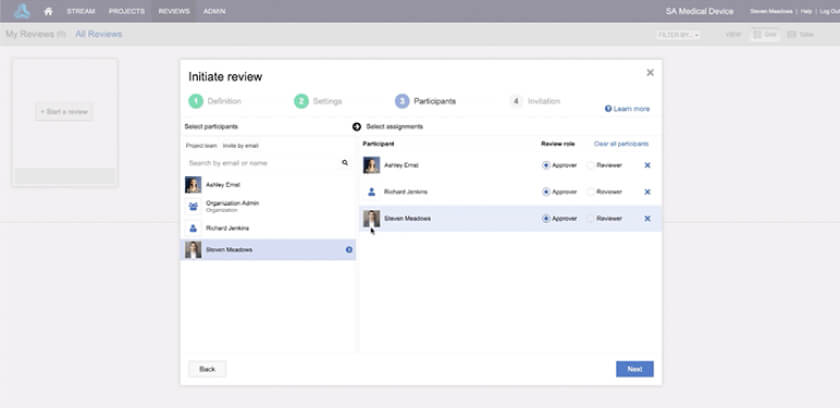
• Increase participation in the review process
• Provide visibility sooner in the review process
• Generate approval-ready content for e-signature faster
Design Trace Matrix is an example of pre-formatted reports used for documentation purposes
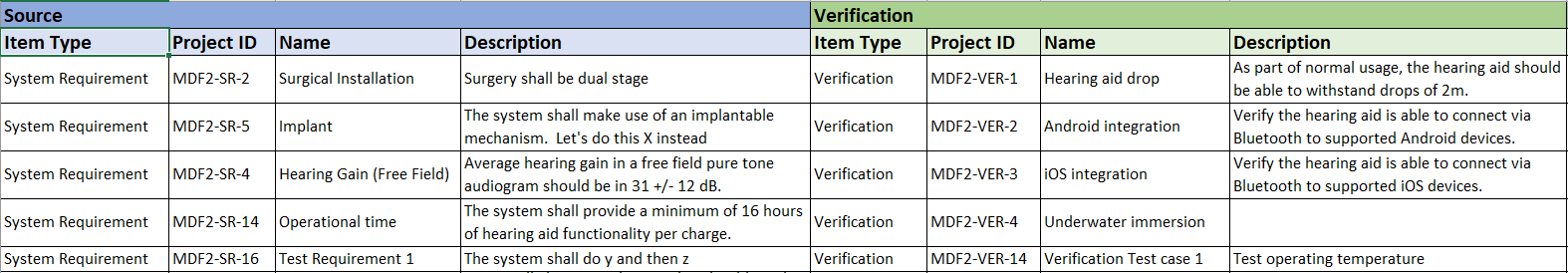
Produce high quality auditable DHF/Technical file documents for your existing quality management system.

Jama Software is the only vendor in the space that offers the following: SOC 2 Type 2 certified application and data center (others data center only), SLAs above 99% (others at 95% or lower), touchless upgrades (others require your IT to manually update hosted app), high availability SLA (others don’t offer), disaster recovery SLA (others don’t offer).
Suitably validated by TÜV SÜD for safety-related development
Jama Software codes with OWASP best practices
Jama Connect is SOC2 Type 2 certified in both the server and application
Ensures strong privacy management practices
Transport Layer Security (TLS) ensures data transferred is secured and encrypted
© Copyright 2000-2025 COGITO SOFTWARE CO.,LTD. All rights reserved